To understand the function of a protein, one must study all of its components. Glycosylation, a covalent attachment of different glycans (oligosaccharides) to the protein backbone, significantly affects glycoprotein stability and function. This dynamic process is regulated by a complex network of genes and involves multiple cellular components, e.g. glycan precursors, sugar transporters, and enzymes involved in glycan attachment and removal, all in the context of their dynamic cellular compartmentalization. Although glycans are involved in many biological processes, analysis of a glycan part of a glycoprotein has often been omitted, mostly due to analytical challenges.
Free iron levels in biological fluids are regulated by transferrin, an iron-binding glycoprotein produced mainly by liver cells. Its glycosylation has been predominantly studied as a biomarker for congenital disorders of glycosylation and chronic alcohol consumption, while inter-individual variability and environmental factors affecting transferrin glycosylation have not been explored in much depth. On the other hand, immunoglobulin G (IgG), the most abundant antibody in human blood plasma, has been extensively studied. Glycosylation of IgG has been recognized to significantly impact its function in the immune system and aberrant patterns have been observed in different diseases.
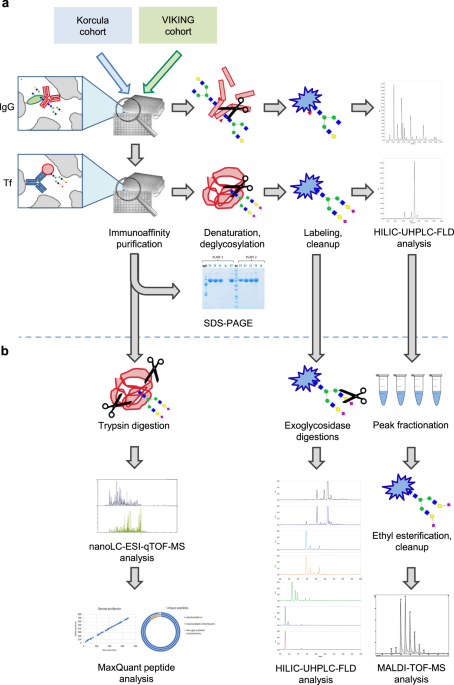
We have developed a highly specific high-throughput approach for the purification of transferrin from blood plasma and its glycosylation analysis. Developed reusable monolithic plate with immobilized antibodies for human transferrin enables its fast large-scale and cost-effective purification without the use of packed columns or tips. Transferrin glycosylation has been characterized based on several complementary analytical approaches expanding on the current knowledge of glycan structures repertoire.
A developed state-of-the-art analytical approach was applied to perform a large-scale comparative study of transferrin and IgG glycosylation in nearly 2000 individuals originating from two populations – Korcula and the Shetland Islands. We demonstrate that both transferrin and IgG glycosylation associate with age, sex, and multiple biochemical and physiological traits, however, with different association patterns. In general, transferrin glycosylation correlates more with sex, than with age, compared to IgG, indicating differential roles of glycosylation on these proteins.
The function of IgG is the recognition of foreign pathogens and activation of an appropriate immune response and it was proposed that it somewhat reflects previous antigen encounters during past infections, so it is conceivable IgG glycosylation correlates more with age, than with sex. The main transferrin function is iron binding and transport, and it is somewhat expected that its glycosylation is well conserved during aging. Furthermore, transferrin synthesis and glycosylation seem to be differentially regulated in females and males due to, e.g. menstrual cycles and hormonal fluctuations, which would be in line with previous studies showing that transferrin concentration and glycosylation change during pregnancy and use of oral contraceptives. Associations of transferrin glycosylation with weight and other clinical and biochemical parameters potentially implicate systemic changes in the iron transport by transferrin within the organism, e.g. after intestinal iron uptake, by iron recycling from iron-containing proteins or iron stores reflected by changes in these physiological and metabolic factors.
The study confirms the importance of glycosylation in distinct physiological functions of transferrin and IgG and represents the basis for future population and clinical studies. Furthermore, it provides additional evidence that glycosylation is tightly regulated in a protein- and cell-specific manner, which confirms our previous finding that glycosylation of different proteins is regulated by different genetic networks.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in