It is now well recognized that Parkinson’s disease patients experience signs and symptoms that go beyond motor changes, and alterations to vision is one of these. Vision changes are typically subtle for a patient to notice but recent advances in technology mean that we can now pick up these changes on a micrometer level. This could prove useful in the quest to develop new disease-modifying medicines for Parkinson’s disease, because to do this well we need biological markers (biomarkers) that are altered in disease and that can track treatment effectiveness. Biomarkers that are simple and inexpensive which can be used in both the laboratory and the clinic best assist drug discovery, making the translation of new drugs from the bench to the bedside easier.
The retina is an outpouching of the brain and a number of Parkinson’s disease hallmarks well documented in the brain can also be found in the retina, including changes to dopamine levels and deposition of alpha-synuclein. To better understand the effect of dopamine depletion in the retina, we employed a MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) mouse model which is known to show a reduction in the number of dopamine producing cells in the substantia nigra. Using this mouse model of Parkinson’s disease we examined whether clinically relevant techniques could detect retinal deficits, and if so whether they could be reversed by applying the gold-standard dopamine treatment, levodopa (L-DOPA).
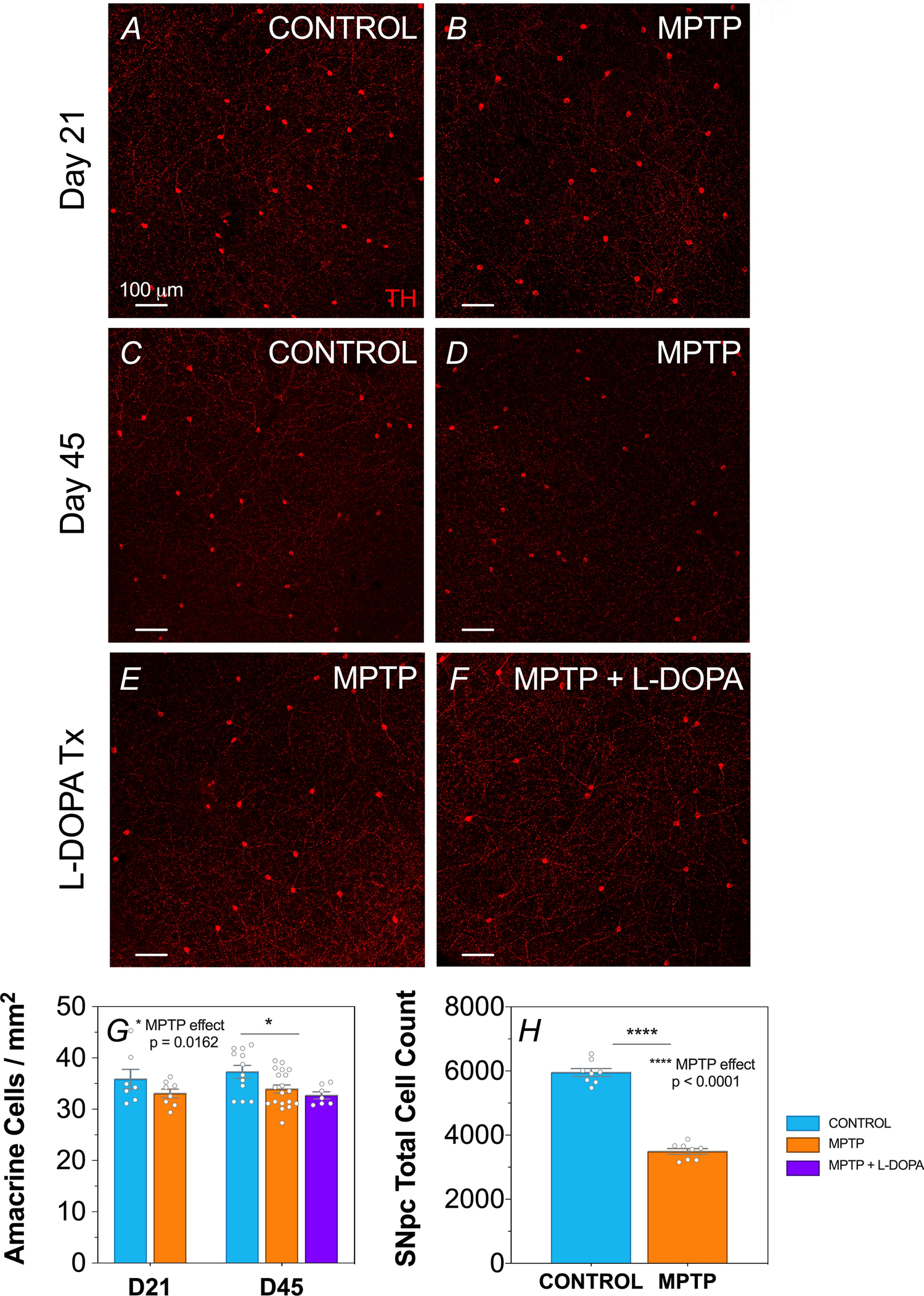
We found that by using a calibrated dose of MPTP (65 mg/kg) we produced the expected loss of cells in the substantia nigra (~60%) and a subtle but significant reduction (~10%) in the number of dopaminergic amacrine cells in the retina. Using in vivo techniques commonly used in eyecare clinics (optical coherence tomography eye-scans and electroretinography) we found retinal changes that were in-agreement with loss of dopaminergic amacrine cells. More specifically, eye-scans revealed specific thinning of the retinal layer where the light collecting photoreceptors communicate to bipolar cells interneurons and dopaminergic amacrine cells, the outer plexiform layer. Using surface electrodes to measure retinal responses to flashes of light (electroretinography) we found a slowing of the oscillatory potentials, which are known to be driven by amacrine cells.
Interestingly, when we treated these Parkinson’s disease mice with L-DOPA, we found that this restored amacrine cell function (speedier and larger oscillatory potential waveforms) but L-DOPA did not improve structural changes as the outer plexiform layer was still thinner and dopaminergic amacrine cells were still lower. Together this suggests that the presence of L-DOPA could temporarily replace lost dopamine but did not treat the underlying Parkinson’s induced deficits.
This study shows that the earliest retinal dopamine-related changes can be detected in a high throughput mouse model of Parkinson’s. These results are in broad agreement with electrophysiology studies in humans originally conducted 35 years ago showing altered electroretinograms in individuals with Parkinson’s disease, that was reversible with L-DOPA. In human eye-scans foveal retinal thinning has been shown in many recent studies, providing a hint that the outer retina may be altered. This study provides insight into the role of dopamine in the retina and points to the need for future research to examine the ocular impact of other Parkinson’s pathologies such as alpha-synuclein.
With eye-scans becoming routine at eyecare clinics and recent advances in portable handheld technologies making electroretinography in humans simpler and less expensive, ocular biomarkers such as those employed in the current study are poised for clinical translation. Biomarkers such as these are desperately needed to support the search for viable disease modifying Parkinson’s treatments, a need that will only increase as our population ages.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in