The benefits of exercise on cardiovascular health are indisputable.1 Expert panels, convened by cardiology societies such as the American College of Cardiology, American Heart Association and European Society of Cardiology 2,3 recommend a minimum of 150 minutes of moderate-intensity activity, or at least 75 minutes of strenuous physical activity per week. However, highly trained endurance athletes often perform way above this recommendation, which often require a sustained 5- to 6- fold increase in cardiac output for a prolonged period. Such intense levels of exercise are known to induce physiological adaptation, with morphological changes in the heart including increases in left ventricular chamber size, wall thickness, and mass, known as “athlete’s heart”.4
Interestingly, different sports have different impacts on cardiac remodeling, as highly trained athletes who participated in a wide variety of sports showed variation in the (regional) location of LV wall thickening (antero-posterior septal wall, apex, or LV free wall). Also, cardiac microstructural (helical) remodeling has been reported in athletes.5 At the molecular level, CITED4 (CBP/p300-interacting transactivators with E [glutamic acid]/D [aspartic acid]-rich-carboxylterminal domain) has been shown to be upregulated in the heart, making it an important transcription factor mediating exercise-induced cardiac growth.6 However, whether the spatial distribution of CITED4 in the heart is associated with (regional) microstructural changes in exercise-induced cardiac hypertrophy remains unclear. Two questions that came to our minds were in which segments (according to American Heart Association segments) CITED4 is expressed and upregulated in the heart following exercise and whether it is correlated with microstructural changes. To bridge this gap, we utilized RNA fluorescence in situ hybridization (RNA-FISH) assay to assess the spatial distribution of CITED4, and diffusion tensor MRI (DT-MRI) to evaluate the microstructural changes in the heart of preclinical animal model of exercise. Of note, DT-MRI is a powerful non-invasive imaging tool to determine small scale fiber orientation changes associated with the disease pathology.
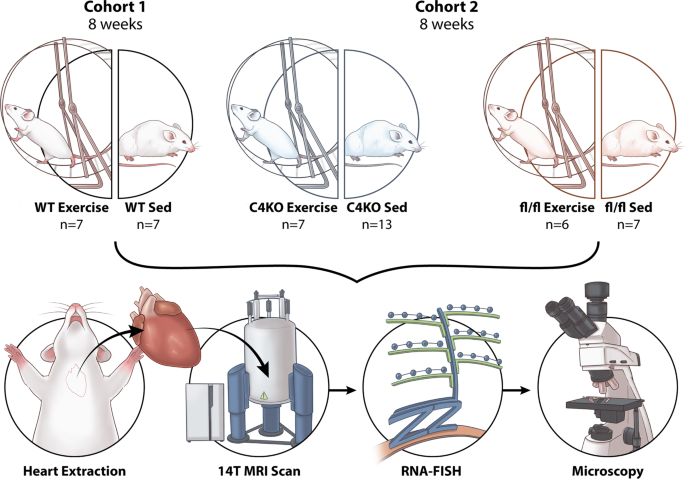
In our initial study, we performed ex-vivo RNA-FISH and DT-MRI on wild type (C57BL/6) mice that were randomized to sedentary or voluntary wheel running for 8 weeks. We observed similar findings with previous reports of increased cardiac CITED4 expression following exercise. Additionally, by using RNA-FISH we were able to analyze the specific spatial expression of CITED4 within the sedentary and exercised mice. We found that the expression of CITED4 was not homogenous across the heart. Rather, CITED4 was specifically upregulated in the lateral wall of the LV in exercised mice. From DTI, we observed that the exercised animals showed changes in their cardiac microstructure through increases in the measure of helicity. These are features frequently reported following exercise and often associated with beneficial remodeling which may lead to a better cardiac function. Intriguingly, we showed that the increases in helicity occurred predominantly in the lateral wall correlating with the increased expression of CITED4.
This finding raised our interest to validate the correlation between CITED4 and cardiac microstructural changes in mice with cardiomyocyte specific CITED4 knockout (KO) mice and their control littermates using similar experimental protocol. After imaging their hearts following 8 weeks of exercise, we did not find the same helical changes present in the wild type mice in the knockout cohort. Interestingly, the groups of mice with CITED4 KO appeared to have baseline helical deficits suggesting that an inability to express CITED4 may have impaired normal cardiac fiber helical structure.
In summary, the novel imaging technologies used in our study are generalizable and can be used to spatially link other molecular mechanisms to microstructural tissue remodeling. Our study also demonstrates that expression of CITED4 following exercise differs by cardiac regions, which may explain the adaptive patterns of cardiac remodeling determined by cardiac DT-MRI. Finally, we further uncover the crucial role CITED4 plays in necessitating these microstructural changes following exercise. Taken together, these results suggest that CITED4 is necessary for beneficial cardiac remodeling to occur following exercise and prompts interesting questions about the role CITED4 plays in the heart’s microstructural changes.
References
- Moreira, J. B. N., Wohlwend, M. & Wisløff, U. Exercise and cardiac health: physiological and molecular insights. Nat. Metab. 2, 829–839 (2020).
- Arnett, D. K. et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 140, (2019).
- Pelliccia, A. et al. 2020 ESC Guidelines on sports cardiology and exercise in patients with cardiovascular disease. Eur. Heart J. 42, 17–96 (2021).
- O’Keefe, J. H. et al. Potential Adverse Cardiovascular Effects From Excessive Endurance Exercise. Mayo Clin. Proc. 87, 587–595 (2012).
- Das, A. et al. Insight Into Myocardial Microstructure of Athletes and Hypertrophic Cardiomyopathy Patients Using Diffusion Tensor Imaging. J. Magn. Reson. Imaging 53, 73–82 (2021).
- Boström, P. et al. C/EBPβ Controls Exercise-Induced Cardiac Growth and Protects against Pathological Cardiac Remodeling. Cell 143, 1072–1083 (2010).






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in